-
Cambrex wraps up expansion of Snapdragon API plant in Massachusetts
- Source: drugdu
- 371
- May 20, 2023
-
Abbott wins FDA spinal cord stimulation approval to challenge Nevro for back pain market
- Source: drugdu
- 372
- May 20, 2023
-
US Appeals Court Panel Questions FDA Due Diligence on Abortion Pill
- Source: drugdu
- 303
- May 20, 2023
-
FDA clears insertable cardiac monitor from Abbott
- Source: drugdu
- 351
- May 20, 2023
-
Pfizer puts kibosh on 4 antibiotics in India after contractor’s manufacturing issues
- Source: drugdu
- 340
- May 20, 2023
-
CDC urges mpox vaccination among people at high risk to prevent summer surge
- Source: drugdu
- 308
- May 20, 2023
-
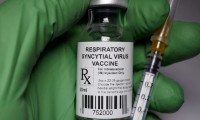
FDA advisors recommend Pfizer’s RSV vaccine for infants but raise safety concerns
- Source: drugdu
- 312
- May 20, 2023
-
Melanin structure discovery brings scientists one step closer to developing ultra-protective sunscreen
- Source: drugdu
- 567
- May 20, 2023
-
Despite Economic Pressures, 90% of Employers Will Continue Wellbeing Programs, Survey Shows
- Source: drugdu
- 304
- May 20, 2023
-
AI voice coach shows promise in psychotherapy for depression and anxiety
- Source: drugdu
- 638
- May 19, 2023
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.