-
FDA Approves Healthy.io Smartphone Camera-based Home Urine Analysis
- Source: MobiHealthNews
- 940
- July 27, 2018
-
Siemens gets FDA Clearance for Troponin Assay
- Source: FierceBiotech
- 1,401
- July 27, 2018
-
Good Feedback for OMS721 Stem Cell Transplant Therapy
- Source: News Medical
- 1,059
- July 27, 2018
-
Enquiry for Nucala in COPD
- Source: BioPharmaDive
- 1,096
- July 26, 2018
-
NHS to Utilize $540 Million New Funding to Boost Hospital IT
- Source: HealthcareIT News
- 1,096
- July 25, 2018
-
Telemedicine Helps Vanderbilt Expand its Transplant Center’s Reach
- Source: mHealth Intelligence
- 1,169
- July 25, 2018
-
Karyopharm Seeks FDA Approval for Selinexor Drug for Multiple Myeloma
- Source: MedCityNews
- 678
- July 24, 2018
-
US Army Awaiting to Test MEDHUB – A Telemedicine Platform
- Source: mHealth Intelligence
- 1,223
- July 23, 2018
-
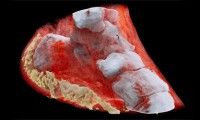
New 3D Color X-Rays to Diagnose Cancer, Heart Disease and More
- Source: Medical Design and Outsourcing
- 1,847
- July 20, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.