-
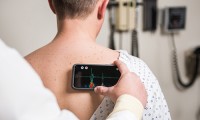
Handheld device from Israeli startup to let pregnant women take smartphone ultrasound scans
- Source: drugdu
- 1,209
- May 11, 2018
-
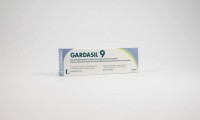
Lynparza tablets (ovarian cancer) given go-ahead by European regulators
- Source: drugdu
- 1,176
- May 10, 2018
-
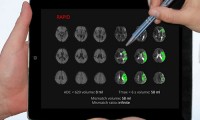
AHA new digital health screening services to identify chronic disease risk
- Source: drugdu
- 1,008
- May 4, 2018
-
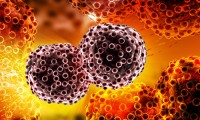
Tisagenlecleucel Granted FDA Approval for Large B-Cell Lymphoma
- Source: targetedonc
- 3,960
- May 3, 2018
-
AbbVie Submits Biologics License Application to U.S. FDA for Investigational Treatment Risankizumab for Moderate to Severe Plaque Psoriasis
- Source: prnewswire
- 1,008
- April 27, 2018
-
FDA to expedite review of IDx-DR, a breakthrough AI diagnostic system
- Source: prweb
- 1,267
- April 26, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.