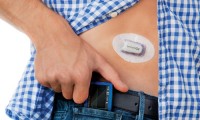
-
FDA clears Medtronic’s smartphone-connected CGM for MDI users
- Source: mobihealthnews
- 1,175
- March 30, 2018
-
FDA clears new Dexcom CGM that requires no patient calibration earlier than expected
- Source: MedCityNews
- 1,312
- March 29, 2018
-
First European filing acceptance of a selective sodium glucose cotransporter-2 (SGLT-2) inhibitor in type-1 diabetes
- Source: astrazeneca
- 1,031
- March 7, 2018
-
Diabetes–New compounds may lower blood sugar but prevent weight gain
- Source: medicalnewstoday
- 1,053
- October 26, 2017
-
Sanofi wins tentative FDA nod for Admelog insulin lispro injection
- Source: yahoo
- 1,088
- September 7, 2017
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.