-
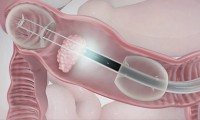
Lumendi Acquires CE-Mark on DiLumen EIP Device
- Source: The Verdict
- 1,617
- July 23, 2018
-
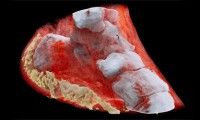
New 3D Color X-Rays to Diagnose Cancer, Heart Disease and More
- Source: Medical Design and Outsourcing
- 1,838
- July 20, 2018
-
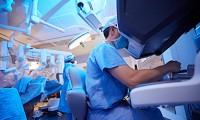
Robotic Surgery – Effective as Traditional Open Surgery Via RAZOR Trial
- Source: The Verge
- 2,351
- July 16, 2018
-
FDA Clearance for Echonous Vein Ultrasound
- Source: The Verge
- 5,910
- July 16, 2018
-
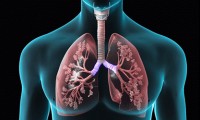
COPD Assessment by CT Analysis
- Source: IndiaTimes
- 2,398
- July 2, 2018
-
Invention of Smartphone Microscope
- Source: IndiaTimes
- 2,334
- June 29, 2018
-
3 Amazing (and Real) Medical Inventions!
- Source: News 24
- 2,478
- June 29, 2018
-
What are the Impacts of Multiple Stents?
- Source: India Times
- 1,329
- June 26, 2018
-
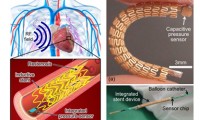
Smart Stent to Continuously Monitor Blood Flow
- Source: ScienceDaily
- 2,027
- June 22, 2018
-
AuriGen Aims to Raise Funds for Development of Irregular Heartbeat Device
- Source: The Independent
- 921
- June 19, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.