-
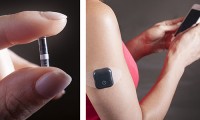
Carewave Medical raising cash for pain relieving wearable, with support from big biotech names
- Source: GeekWire
- 1,045
- February 28, 2018
-
Implanted continuous glucose sensor proven safe
- Source: globalbiotechinsights
- 1,248
- February 28, 2018
-
Samsung’s Galaxy S9 to include blood pressure monitor, research app
- Source: mobihealthnews
- 983
- February 28, 2018
-
Kardian’s contactless monitoring system detects falls
- Source: mobihealthnews
- 968
- February 27, 2018
-
Final FDA Rule Requires Medical Device Trials Outside US to Conform to GCP
- Source: Regulatory Affairs Professionals Society
- 1,203
- February 24, 2018
-
CFDA and CMDE Issue Draft Guidance on Medical Devices for Rare Diseases
- Source: National Law Review
- 1,114
- February 23, 2018
-
Medical device product development– 7 pitfalls you need to avoid
- Source: medicaldesignandoutsourcing
- 970
- February 14, 2018
-
4 causes of failure for medical devices
- Source: multibriefs
- 1,124
- February 12, 2018
-
Drugdu.com (Ddu)– Professional Platform for Cross-border Medical Trade
- Source: Ddu
- 1,795
- February 9, 2018
-
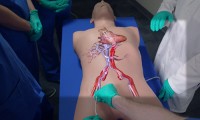
Surgeons use Microsoft HoloLens to ‘see inside’ patients before they operate on them
- Source: Microsoft
- 1,092
- February 9, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.