-
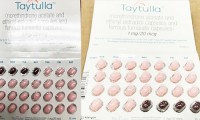
Packaging Error Prompts Allergan to Recall Sample Packs of Taytulla Contraceptive Pills
- Source: Ddu
- 1,233
- May 30, 2018
-
Healthcare Sector Has Not Fully Exploited Data Analytics Potential
- Source: Ddu
- 1,142
- May 28, 2018
-
Sesen Bio Presents Bladder Cancer Treatment Data Following Name Change
- Source: Ddu
- 1,158
- May 22, 2018
-
Security issues with Philips, GE, and Silex medical devices
- Source: HealthIT news
- 1,025
- May 15, 2018
-
Integration of next-generation sequencing data in clinical trials facilitated by DNAnexus
- Source: Ddu
- 830
- May 15, 2018
-
Qventus raised $30M investment from Bessemer Venture Partners
- Source: Ddu
- 816
- May 15, 2018
-
Biotechnology Company Abpro postpones $69M IPO
- Source: Ddu
- 860
- May 14, 2018
-
Amazon focusing to upend healthcare sector with Alexa
- Source: Ddu
- 1,026
- May 11, 2018
-
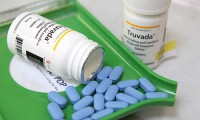
Safer HIV drugs taken off the shelf by Gilead
- Source: drugdu
- 1,188
- May 11, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.