-
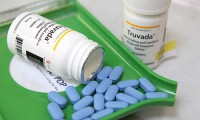
Safer HIV drugs taken off the shelf by Gilead
- Source: drugdu
- 1,189
- May 11, 2018
-
Gilead inks $90m deal with Verily to create “molecular map” of inflammatory disease
- Source: drugdeliverybusiness
- 974
- May 2, 2018
-
Gilead links with Sangamo in gene therapy alliance
- Source: drugdu
- 899
- February 26, 2018
-
J&J lines up its 4-in-1 HIV pill for FDA behind rivals from Gilead, GSK
- Source: Endpts
- 1,010
- September 27, 2017
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.