-
Researchers Identify Genes that Directly Influence What We Eat
- Source: drugdu
- 495
- July 24, 2023
-
U.S. Healthcare Pandemic Recovery Requires Focused Support of Independent Primary Care
- Source: drugdu
- 306
- July 24, 2023
-
Immune systems develop specific genes to fight against common bacteria, study finds
- Source: drugdu
- 525
- July 22, 2023
-
New ultra-flexible neural implant records single-neuron activity in deep-brain regions
- Source: drugdu
- 555
- July 22, 2023
-
Novartis acquires DTx Pharma in deal worth up to $1bn
- Source: drugdu
- 379
- July 22, 2023
-
AstraZeneca/Sanofi’s RSV antibody receives FDA approval for use in infants
- Source: drugdu
- 307
- July 22, 2023
-
Sandoz doubles down on biosimilars with $90M Slovenia site where it plans to hire 200
- Source: drugdu
- 391
- July 22, 2023
-
WHO director-general calls out critics of planned pandemic accords
- Source: drugdu
- 292
- July 22, 2023
-
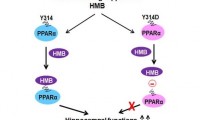
Bodybuilding supplement may help stave off progression of Alzheimer’s
- Source: drugdu
- 398
- July 22, 2023
-
Sanofi Puts Up $125M to Partner on Oral Drug That Could Be Dupixent’s Successor
- Source: drugdu
- 549
- July 22, 2023
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.