-
Bavarian Nordic locks up $120M BARDA deal to replenish US stock of smallpox, mpox vaccine
- Source: drugdu
- 711
- August 7, 2023
-
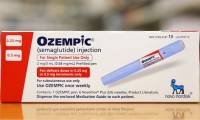
Novo Nordisk, Eli Lilly face injury lawsuit from user of popular GLP-1 medicines
- Source: drugdu
- 368
- August 7, 2023
-
GSK files lawsuit against Pfizer over RSV vaccine patent infringement
- Source: drugdu
- 500
- August 7, 2023
-
NICE publishes draft guidance not recommending CSL’s haemophilia B gene therapy
- Source: drugdu
- 380
- August 7, 2023
-
With Takeda and Hutchmed hot on the trail, Taiho wins FDA nod for stronger Lonsurf regimen in colorectal cancer
- Source: drugdu
- 438
- August 4, 2023
-
Augmedics picks up Surgalign’s digital health assets for $900K
- Source: drugdu
- 446
- August 4, 2023
-
Liquid biopsy may be used to identify gene mutations in pediatric arteriovenous malformations
- Source: drugdu
- 502
- August 4, 2023
-
Scleroderma Research Foundation launches clinical trial platform to advance treatments
- Source: drugdu
- 769
- August 4, 2023
-
Amygdala scores $2m NIAAA grant to develop alcohol use disorder therapy
- Source: drugdu
- 455
- August 4, 2023
-
CS Pharmaceuticals snaps up AxeroVision to shore up ophthalmology portfolio
- Source: drugdu
- 386
- August 4, 2023
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.