-
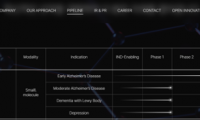
$770 Million! AriBio authorizes Marketing Rights for a Drug to Treat Alzheimer’s Disease in China
- Source: drugdu
- 384
- March 28, 2024
-
EU recommendations for 2024/2025 seasonal flu vaccine composition
- Source: drugdu
- 815
- March 28, 2024
-
Another Chiatai Tianqing’s contrast agent, iopromide injection, was approved for marketing
- Source: drugdu
- 424
- March 28, 2024
-
BIO-THERA’s Phase II Clinical Trial Application for BAT8006 (ADC- FRα) for Injection Approved by US FDA
- Source: drugdu
- 696
- March 28, 2024
-
Inizio Engage and Applied Driving partner to support clinical workers with safety app
- Source: drugdu
- 511
- March 28, 2024
-
Researchers identify group of biological markers found in high levels in TB patients
- Source: drugdu
- 492
- March 28, 2024
-
Hong Kong Adventist Healthcare and Fosun Health Sign Strategic Cooperation Agreement
- Source: drugdu
- 441
- March 27, 2024
-
Hengrui’s Thiotepa for Injection for Pre-treatment of Hematopoietic Stem Cell Transplantation Approved for Marketing
- Source: drugdu
- 427
- March 27, 2024
-
CanSinoBio’s Quadrivalent Conjugate Vaccine Launches Phase III Clinical Trial in Indonesia
- Source: drugdu
- 482
- March 27, 2024
-
CSPC obtains clinical approval for semaglutide weight loss indication
- Source: drugdu
- 377
- March 27, 2024
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.