-
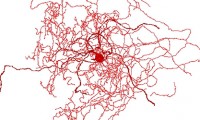
Discovery of Rosehip Neurons in Humans
- Source: FierceBiotech
- 1,422
- August 29, 2018
-
Mersana’s Lead Cancer ADC Held Up by FDA for Patient’s Death
- Source: FierceBiotech
- 589
- July 25, 2018
-
Luspatercept Passes Phase 3 Clinical Trial for Rare Blood Disease
- Source: FierceBiotech
- 1,104
- July 3, 2018
-
Probiotics for Liver Health
- Source: MedicalXpress; Healthline
- 1,274
- June 15, 2018
-
Strong Growth Potential in Global Clinical Trial Data Management and eClinical Solutions Market to Cross $4 Billion by 2020
- Source: PR Newswire
- 972
- April 12, 2018
-
Leading CROs, Veeva team up for open tech on trials
- Source: fiercebiotech
- 1,175
- March 30, 2018
-
ViiV kicks off PhIII trial of two-drug HIV regimen
- Source: pharmatimes
- 1,090
- February 12, 2018
-
Genentech’s Tecentriq Impresses Again in Late-Stage Kidney Cancer Trial
- Source: Biospace
- 1,205
- February 11, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.