-
J&J reportedly eyeing Shockwave Medical for takeover
- Source: drugdu
- 52
- April 2, 2024
-
Hengrui’s Clinical Application for New Drug Indicated for Psoriasis Approved
- Source: drugdu
- 42
- April 2, 2024
-
FDA Approves Akebia’s Vafseo for Anemia in Adults with Chronic Kidney Disease on Dialysis
- Source: drugdu
- 59
- March 30, 2024
-
Boundless Bio’s ‘BOLD’ IPO Reels In $100M for a New Kind of Cancer Drug
- Source: drugdu
- 76
- March 30, 2024
-
Jacobio Pharma Announces 2023 Annual Results
- Source: drugdu
- 49
- March 30, 2024
-
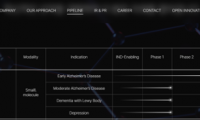
$770 Million! AriBio authorizes Marketing Rights for a Drug to Treat Alzheimer’s Disease in China
- Source: drugdu
- 54
- March 28, 2024
-
BIO-THERA’s Phase II Clinical Trial Application for BAT8006 (ADC- FRα) for Injection Approved by US FDA
- Source: drugdu
- 51
- March 28, 2024
-
FDA Fast Tracks ImmVira’s Oncolytic Herpes Simplex Virus Therapy for Head and Neck Squamous Cell Cancer
- Source: drugdu
- 46
- March 25, 2024
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.