-
Researchers develop new technology platform for cancer immunotherapy
- Source: immunotherapies
- 470
- February 27, 2018
-
Merck Drops $394 Million to Acquire Virus-Based Cancer Company Viralytics
- Source: Biospace;
- 677
- February 26, 2018
-
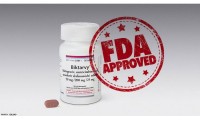
U.S. Food and Drug Administration Approves Gilead’s Biktarvy® (Bictegravir, Emtricitabine, Tenofovir Alafenamide) for Treatment of HIV-1 Infection
- Source: gilead
- 855
- February 26, 2018
-
Novartis receives FDA approval for Cosentyx® label update to include moderate to severe scalp psoriasis
- Source: pharma.us.novartis
- 503
- February 13, 2018
-
Sage Therapeutics Receives FDA Breakthrough Therapy Designation for SAGE-217 for the Treatment of Major Depressive Disorder
- Source: finance.yahoo
- 696
- February 12, 2018
-
Genentech’s Tecentriq Impresses Again in Late-Stage Kidney Cancer Trial
- Source: Biospace
- 597
- February 11, 2018
-
U.S. Food and Drug Administration Approves Gilead’s Biktarvy®
- Source: Finchannel
- 745
- February 9, 2018
-
Why AstraZeneca’s New Drug Strategy Seems to be Bearing Fruit
- Source: china-pharmacy
- 456
- February 7, 2018
-
Seattle Genetics Snaps Up Cascadian in $614M Buyout Deal
- Source: Biospace
- 417
- February 1, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.