-
US announces first ten drugs up for Medicare price negotiation
- Source: drugdu
- 103
- August 31, 2023
-
FTC pulls back on legal challenge to Amgen’s $28B bid for Horizon
- Source: drugdu
- 115
- August 30, 2023
-
AstraZeneca Latest to Sue Biden Administration Over IRA’s Drug Price Negotiations
- Source: drugdu
- 108
- August 30, 2023
-
BridgeBio Builds ATTR-CM Case for Acoramidis with Promising Phase III Data
- Source: drugdu
- 108
- August 30, 2023
-
Moderna says new Covid vaccine was effective against Eris variant in early trial
- Source: drugdu
- 108
- August 20, 2023
-
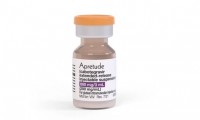
MSF, trying to secure supplies of ViiV’s Apretude, calls out new clauses in purchasing contract
- Source: drugdu
- 135
- August 19, 2023
-
GSK stays a step ahead in RSV vaccine battle as Arexvy becomes available at US pharmacies
- Source: drugdu
- 173
- August 19, 2023
-
Seagen Touts Phase III Data for Breast Cancer Combo Therapy
- Source: drugdu
- 204
- August 18, 2023
-
Arcturus JV scored $115M in Japanese grants to boost mRNA manufacturing
- Source: drugdu
- 231
- August 16, 2023
-
FDA Approves J&J’s Akeega for BRCA-Positive Prostate Cancer
- Source: drugdu
- 187
- August 16, 2023
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.