-
Gilead links with Sangamo in gene therapy alliance
- Source: drugdu
- 498
- February 26, 2018
-
How Precision Medicine is breaking off Chokehold on Healthcare with Big Data
- Source: dexlabanalytics
- 493
- February 23, 2018
-
NICE sticks with ‘no’ for earlier use of Halaven
- Source: pharmatimes
- 1,775
- February 23, 2018
-
Could AI One Day Detect the Flu…Before You Even Feel Sick?
- Source: smithsonianmag
- 557
- February 14, 2018
-
Stanford spin-out Zenflow raises $31M to join the megamarket for enlarged prostate
- Source: MedCityNews
- 641
- February 11, 2018
-
chemoWave app offers patients support during chemotherapy
- Source: MobiHealthNews
- 529
- February 11, 2018
-
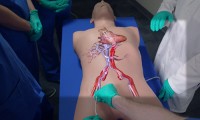
Surgeons use Microsoft HoloLens to ‘see inside’ patients before they operate on them
- Source: Microsoft
- 696
- February 9, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.