-
metaLinear Secures 255K Pound Seed Investment for Targeted Antibacterial Research
- Source: biocity.co.uk;
- 431
- December 1, 2017
-
Prometic Reports Positive Interim Clinical Data From Ongoing Intravenous Immunoglobulin Pivotal Phase III Trial
- Source: Prometic
- 459
- November 28, 2017
-
Massachusetts’ Entasis Snags $31.9 Million To Advance Anti-infective Therapeutic Portfolio
- Source: biospace
- 447
- November 23, 2017
-
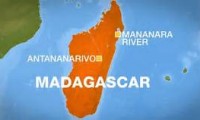
DEATH ROW Madagascar plague doctors say infections have reached ‘crisis point’ as Black Death toll hits 195
- Source: thesun.co.uk
- 392
- November 23, 2017
-
Plague – Madagascar
- Source: who.int
- 383
- November 15, 2017
-
Teva looks to sell generics in China through joint venture with Guangzhou Pharma
- Source: Fiercepharma
- 471
- November 7, 2017
-
China’s Antimicrobial Drug Import and Export Market Report
- Source: Ddu
- 1,307
- October 13, 2017
-
FDA awards 15 grants for clinical trials to stimulate product development for rare diseases
- Source: fda.gov
- 570
- October 9, 2017
-
VED – Vaccines for Enteric Diseases
- Source: medical-events
- 539
- October 3, 2017
-
Two Scientists Nab Another $20 Million for Their CRISPR Biotech Eligo Bioscience
- Source: Biospace
- 483
- September 29, 2017
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.