World’s first! Amgen/AstraZeneca’s blockbuster drug approved for marketing in China
March 31, 2026
Source: drugdu
 155
155


On March 27, the NMPA website showed that terzafimumab injection, a TSLP monoclonal antibody jointly submitted by AstraZeneca and Amgen , was approved for marketing. The drug was approved for two indications: asthma (application number: JXSS2400102/3) and chronic sinusitis with nasal polyps (application number: JXSS2500019/20) .


Screenshot source: NMPA official websiteTezaliumab, developed in collaboration between AstraZeneca and Amgen, is a first-in-class human monoclonal antibody that inhibits the action of TSLP. TSLP is a key epithelial cytokine located at the top of multiple inflammatory cascades and is closely associated with allergic, eosinophilic, and other types of endothelial inflammation related to severe asthma and other inflammatory diseases.In 2021, the drug was first approved by the FDA for adjunctive maintenance therapy in adults and children aged 12 years and older with severe asthma . In 2022, it was approved in the European Union and Japan. In October 2025, the drug was approved for a new indication in the United States for maintenance therapy of poorly controlled chronic sinusitis with nasal polyps (CRSwNP) in adults aged 12 years and older and children.
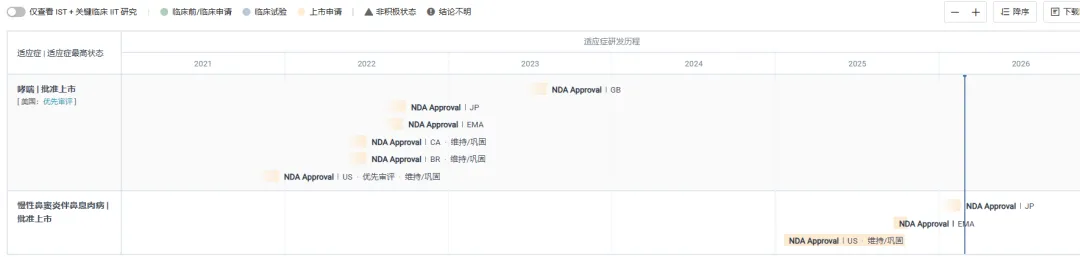
In China, the drug has initiated Phase III clinical trials for asthma , eosinophilic esophagitis , chronic sinusitis with nasal polyps, and chronic obstructive pulmonary disease .
asthma
A DESTINATION clinical trial on asthma has been completed. This was a regional, multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase III study designed to evaluate the efficacy and safety of tesazelumab in adult subjects with poorly controlled severe asthma.
Clinical results presented at the 2025 American Academy of Allergy, Asthma & Immunology (AAAAI) Annual Meeting showed that tesserumab significantly reduced the rate of acute asthma exacerbations (AAER) by 74% over 52 weeks compared to placebo .
Regarding secondary endpoints, compared with placebo, tesalimumab significantly improved lung function over 52 weeks, with an improvement of FEV1 of 250 ml, and significantly improved asthma symptom control and quality of life (based on ACQ-6, AQLQ[S]+12 and ASD assessments, all of which showed significant improvement from baseline after 52 weeks of treatment) .
Chronic sinusitis with nasal polyps
Positive results from the Phase III WAYPOINT clinical trial demonstrated that, compared with placebo, teserlimumab significantly reduced the severity of nasal polyps in patients with chronic sinusitis and nasal polyps, reducing the need for subsequent nasal polyp surgery and systemic corticosteroid use . The relevant data were published in the *New England Journal of Medicine* and will be presented as an oral presentation of a groundbreaking study at the 2025 AAAAI Congress.
Data showed that tesalimumab treatment significantly reduced the severity of nasal polyps as assessed by the co-primary endpoint: compared with placebo, at week 52, the nasal polyp score (NPS) decreased by 2.065 points and the patient-reported nasal congestion symptom score (NCS) decreased by 1.028 points . Improvements in NPS were first observed at week 4, and improvements in NCS were first observed at week 2 (both post-treatment assessments) , with efficacy persisting until week 52.
In the overall study population, all key secondary endpoints showed statistically and clinically significant improvements. Most importantly, compared to the placebo group, teserlimumab significantly reduced the need for subsequent nasal polyp surgery (by up to 98%) and the need for systemic glucocorticoid use (by up to 88%).
Insight Perspective
According to Insight database, tesalimumab is currently the only TSLP monoclonal antibody approved for marketing globally , with sales approaching $2 billion in 2025, making its debut on the list of the top 100 global pharmaceutical sales . In addition, six other drugs have entered Phase III clinical trials, from companies such as Biosin/Chia Tai Tianqing (Bosakitug) , Hengrui/GSK (GSK5784283) , Connoya/CSPC (CM326) , and Quanxin Biotech (JKN24011) . Among them, Qilu Pharmaceutical's QL2302 is a tesalimumab biosimilar.

Disclaimer:
The content of this article is for reference only and does not constitute investment advice. Investors act at their own risk. We remain neutral on the statements and opinions expressed in this article and provide no express or implied warranty as to the accuracy, reliability, or completeness of the content. Readers should use this information for reference only and assume full responsibility for their own actions.
https://mp.weixin.qq.com/s/JYs0t7J2oB-SiYu1QJobXw
By editor
Read more on
- Cinda leads the world, and Takeda’s 1.2 billion gamble is reaping the rewards. June 5, 2026
- The National Medical Products Administration (NMPA) conditionally approved the marketing of Pometamethasone Alpha for injection. June 5, 2026
- The generic version of this drug, developed by 35 pharmaceutical companies, has had its market launch delayed by 5 years. June 5, 2026
- Pharmaceutical giant lays off more employees June 5, 2026
- Imported “new kidney disease drug” approved June 5, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



