Juventas Biotech’s third new drug application! The new drug application for YuanRuida, used to treat childhood acute lymphoblastic leukemia, has been accepted
May 14, 2026
Source: drugdu
 105
105
 On May 11 , 2026 , Juventas Biotechnology Co., Ltd. (hereinafter referred to as " Juventas Biotechnology " ) announced that the National Medical Products Administration ( NMPA ) has formally accepted its New Drug Application ( NDA ) for its core product , YuanRuida® (Nakiolensai Injection) , for the treatment of relapsed or refractory B- cell acute lymphoblastic leukemia ( r/r B-ALL ) in children. This is the third New Drug Application submitted by Juventas Biotechnology, a Chinese-developed CD19 CAR-T product , following the NMPA approvals for its two other New Drug Applications for the treatment of relapsed or refractory B- cell acute lymphoblastic leukemia in adults and relapsed or refractory large B- cell lymphoma in adults . This marks another significant milestone for Juventas Biotechnology in the research and development of innovative CAR-T cell drugs. YuanRuida® becomes the only CAR-T product capable of simultaneously covering adult acute lymphoblastic leukemia, childhood acute lymphoblastic leukemia, and large B- cell lymphoma , bringing breakthrough treatment options to more patients.
On May 11 , 2026 , Juventas Biotechnology Co., Ltd. (hereinafter referred to as " Juventas Biotechnology " ) announced that the National Medical Products Administration ( NMPA ) has formally accepted its New Drug Application ( NDA ) for its core product , YuanRuida® (Nakiolensai Injection) , for the treatment of relapsed or refractory B- cell acute lymphoblastic leukemia ( r/r B-ALL ) in children. This is the third New Drug Application submitted by Juventas Biotechnology, a Chinese-developed CD19 CAR-T product , following the NMPA approvals for its two other New Drug Applications for the treatment of relapsed or refractory B- cell acute lymphoblastic leukemia in adults and relapsed or refractory large B- cell lymphoma in adults . This marks another significant milestone for Juventas Biotechnology in the research and development of innovative CAR-T cell drugs. YuanRuida® becomes the only CAR-T product capable of simultaneously covering adult acute lymphoblastic leukemia, childhood acute lymphoblastic leukemia, and large B- cell lymphoma , bringing breakthrough treatment options to more patients.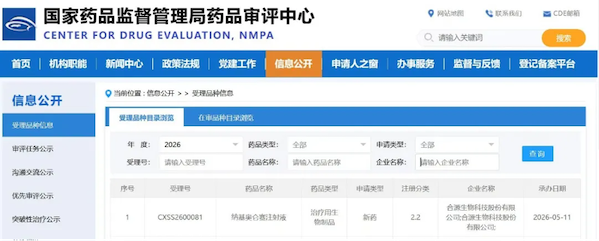 Acute lymphoblastic leukemia ( ALL ) is the most common hematologic malignancy in children, accounting for more than 70% of childhood leukemia cases . Although combination chemotherapy and precise stratification therapy have improved the overall survival rate of children with ALL to 80%-90% , 20%-25% of patients still experience relapse or primary refractory disease. This group has a very poor prognosis, with a salvage success rate of only 31% for primary refractory patients and a median overall survival ( OS ) of less than one year. As the number of treatment lines increases, the remission rate in pediatric patients continues to decline, and the severe side effects of long-term multi-drug chemotherapy, such as serious infections, bone marrow suppression, and organ toxicity, severely impact the quality of life and long-term prognosis of these children. Therefore, there is an urgent clinical need for safe and effective innovative treatment methods.
Acute lymphoblastic leukemia ( ALL ) is the most common hematologic malignancy in children, accounting for more than 70% of childhood leukemia cases . Although combination chemotherapy and precise stratification therapy have improved the overall survival rate of children with ALL to 80%-90% , 20%-25% of patients still experience relapse or primary refractory disease. This group has a very poor prognosis, with a salvage success rate of only 31% for primary refractory patients and a median overall survival ( OS ) of less than one year. As the number of treatment lines increases, the remission rate in pediatric patients continues to decline, and the severe side effects of long-term multi-drug chemotherapy, such as serious infections, bone marrow suppression, and organ toxicity, severely impact the quality of life and long-term prognosis of these children. Therefore, there is an urgent clinical need for safe and effective innovative treatment methods.
Yuanruida® (Nakiolensai Injection) is a CD19-targeted CAR-T cell therapy product developed by Juventas Biotechnology with completely independent intellectual property rights . It possesses a globally unique CD19 scFv ( HI19a ) structure and internationally leading manufacturing processes, demonstrating consistently excellent efficacy and safety in both registration clinical studies and real-world trials. Previously, Yuanruida® received approval from the National Medical Products Administration ( NMPA ) in November 2023 for the treatment of adult relapsed or refractory B- cell acute lymphoblastic leukemia, making it the only CAR-T product in China specifically for treating adult acute lymphoblastic leukemia . In November 2025 , Yuanruida® received its second new drug approval for the treatment of adult relapsed or refractory large B- cell lymphoma, becoming the only CAR- T product in China simultaneously covering both leukemia and lymphoma, two major hematological malignancies .
Yuanruida® (Nakiolensai Injection) for the treatment of relapsed or refractory B-cell acute lymphoblastic leukemia in children received " Breakthrough Therapy " designation from the Center for Drug Evaluation ( CDE ) of the National Medical Products Administration (NMPA) in August 2025 , enjoying accelerated policy support such as priority review. On May 11 , 2026 , the new drug application for pediatric acute lymphoblastic leukemia was officially accepted by the NMPA . Based on its excellent efficacy and controllable safety demonstrated in the registration clinical trial, it is expected to be approved for marketing quickly.
Data from a pivotal registration clinical trial ( Phase I/II , multicenter, single-arm, open-label) of nagiolenza injection in the treatment of recurrent/rheumatoid arthritis (r/r B-ALL ) in children showed the following: High objective response rate ( ORR ): The ORR in evaluable patients reached 90% , with a complete response ( CR ) rate exceeding 70% , significantly superior to traditional salvage chemotherapy; Deep MRD negativity: All remission patients achieved minimal residual disease ( MRD ) negativity, with a 100% MRD negativity rate , laying a core foundation for long-term disease-free survival . Rapid remission and durable benefit: All remission patients achieved MRD negativity at the first assessment 28 days after cell infusion , and some children have achieved more than 2 years of sustained disease-free survival, returning to normal life and school . Adverse reactions were mainly grade 1-2 cytokine release syndrome ( CRS ) , with a low incidence of grade ≥3 CRS and no treatment-related deaths; the incidence of immune effector cell-related neurotoxicity syndrome ( ICANS ) was low, and all patients recovered without sequelae after clinical treatment. The overall safety profile was consistent with that of adult patients and met the requirements for pediatric treatment tolerability .
About Juventas Bio
Founded in June 2018, Juventas Bio has grown into a leader in independent innovation in China's cell therapy industry, committed to becoming a globally leading next-generation biopharmaceutical company driven by innovative cell and gene technologies. The company's first core product, YuanRuida® (Nanaki Olensai Injection, CNCT19), was officially approved for marketing by the National Medical Products Administration in November 2023. This product is the first CAR-T drug in China's
https://mp.weixin.qq.com/s/Fg8qXR2YzFIRgXJIgIrvrw
By editorRead more on
- Cinda leads the world, and Takeda’s 1.2 billion gamble is reaping the rewards. June 5, 2026
- The National Medical Products Administration (NMPA) conditionally approved the marketing of Pometamethasone Alpha for injection. June 5, 2026
- The generic version of this drug, developed by 35 pharmaceutical companies, has had its market launch delayed by 5 years. June 5, 2026
- Pharmaceutical giant lays off more employees June 5, 2026
- Imported “new kidney disease drug” approved June 5, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



