Chongqing Yaoyou Pharmaceutical Receives Approval for 2 Billion Yuan Worth of Injectable Drugs
March 19, 2026
Source: drugdu
 89
89

Moxifloxacin hydrochloride sodium chloride injection is an 8-methoxyfluoroquinolone antibiotic with broad-spectrum activity and bactericidal effects. It is suitable for acute bacterial sinusitis, acute exacerbations of chronic bronchitis, community-acquired pneumonia, complicated/uncomplicated skin and skin tissue infections, and complicated intra-abdominal infections caused by susceptible bacteria.
According to data from Menet.com , Moxifloxacin Hydrochloride Sodium Chloride Injection is a Class B drug covered by the National Medical Insurance and a National Essential Medicines List. In 2024, its sales in China's three major terminals and six major markets approached 2 billion yuan, recovering to the level before the implementation of the national centralized procurement policy. However, it experienced a certain degree of decline in the first three quarters of 2025.
Sales of Moxifloxacin Hydrochloride Sodium Chloride Injection in China's Three Major Terminals and Six Major Markets in Recent Years (Unit: RMB 10,000)
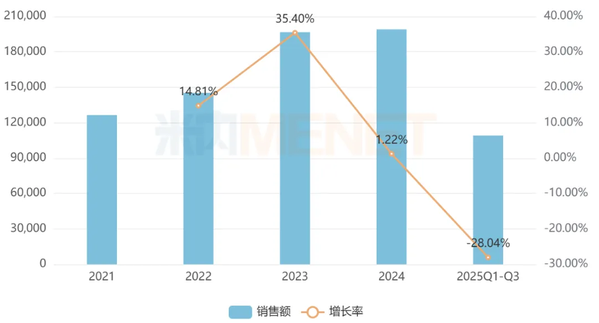 Source: Menet Pattern Database
Source: Menet Pattern Database
Currently, more than 30 domestic companies have passed the evaluation or are deemed to have passed the evaluation for moxifloxacin hydrochloride sodium chloride injection, including CR Double-Crane Pharmaceutical, Nanjing Chia Tai Tianqing Pharmaceutical, CSPC Ouyi Pharmaceutical, Yangtze River Pharmaceutical, Shijiazhuang No.4 Pharmaceutical, Hongri Pharmaceutical, Chongqing Yaoyou Pharmaceutical, etc.
According to data from Menet.com, Chongqing Yaoyou Pharmaceutical currently has 20 systemic anti-infective drugs that have passed or are considered to have passed the evaluation, mainly systemic antibacterial drugs. Among them, isapamicin sulfate injection and clindamycin hydrochloride capsules are the first or only drugs to pass the evaluation.
Systemic anti-infective drugs from Chongqing Yaoyou Pharmaceutical Co., Ltd. that have passed evaluation
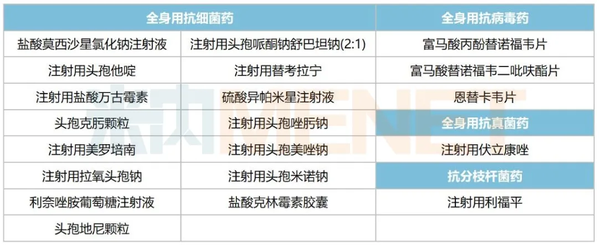 Source: Menet Consistency Evaluation Progress Database
Source: Menet Consistency Evaluation Progress Database
Since 2025, Chongqing Yaoyou Pharmaceutical has had 29 products approved for production in China and deemed to have passed the evaluation. These products are concentrated in major therapeutic categories such as systemic anti-infective drugs, nervous system drugs, anti-tumor drugs, and immunomodulators. Among them are several best-selling products such as moxifloxacin hydrochloride sodium chloride injection, amlodipine besylate tablets, rosuvastatin calcium tablets, clopidogrel bisulfate tablets, atorvastatin calcium tablets, and metformin hydrochloride extended-release tablets.
Products approved for production by Chongqing Yaoyou Pharmaceutical since 2025 and considered as having passed the evaluation
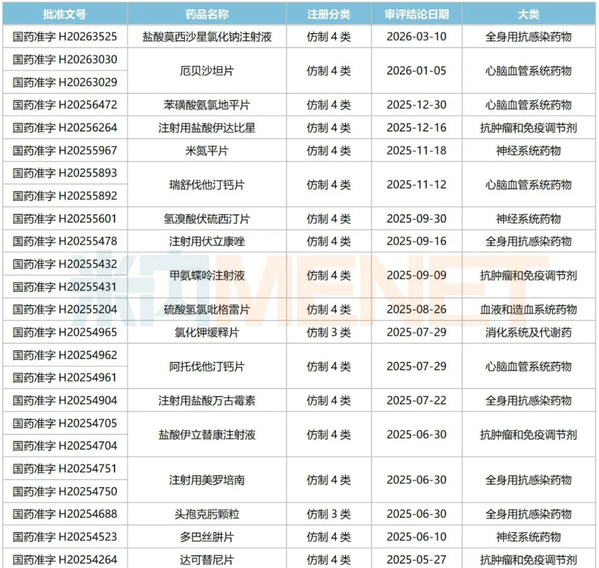
https://mp.weixin.qq.com/s/MjzsPw_hizUqxatjV4xdHA
By editorRead more on
- Boehringer Ingelheim’s new oral drug “Zonaitinib” has been approved for a new indication in China May 22, 2026
- Class 1 innovative drug Fazanleisen tablets approved May 22, 2026
- Shijiazhuang Pharmaceutical Group’s budesonide enteric-coated capsules approved! This poses a challenge to generic drug manufacturers, weakening the original drug’s competitive advantage. May 22, 2026
- Two world-first drugs from Chipscreen Biosciences have been launched in the Macau market, further improving patient accessibility! May 22, 2026
- Beimei Prostol Eye Drops Receives Drug Registration Certificate May 22, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



