A New Era Dawns
February 26, 2026
Source: drugdu
 150
150
The anticoagulation field has given rise to global blockbuster drugs. For instance, apixaban generated global sales of over $20 billion in 2025. Following the iterations of warfarin and non vitamin K antagonist oral anticoagulants (NOACs), the direction for next generation anticoagulants is now clear: FXI/FXIa inhibitors are set to become an entirely new option in anticoagulation therapy.
Recently, Bayer also announced that its FXIa inhibitor asundexian achieved breakthrough positive results in pivotal Phase III clinical trials.The era of FXI/FXIa inhibitors has officially begun.
01Breaking the Deadlock
Thrombotic diseases are a group of disorders characterized by the formation of pathological blood clots in arteries or veins. They have become one of the leading global causes of death, directly linked to roughly one quarter of all deaths worldwide each year.
According to Frost & Sullivan data, the global number of incident cases of thrombotic diseases reached 26.7 million in 2024, with 7 million in China. By 2034, the global figure is projected to rise to 29.1 million, and the patient population in China will climb to 12.4 million. Thrombosis related conditions such as myocardial infarction, ischemic stroke and venous thromboembolism continue to add to the global disease burden. Anticoagulation therapy serves as the cornerstone for the prevention and treatment of thromboembolism, widely used in core clinical scenarios including deep vein thrombosis management, stroke prevention in atrial fibrillation, and anticoagulation for valvular heart disease.
Looking back at the history of anticoagulants, vitamin K antagonists represented by warfarin dominated clinical practice for nearly half a century. However, these agents could not meet refined clinical needs due to inherent drawbacks: large interindividual variability, complex drug food interactions, a narrow therapeutic window, and high bleeding risk.
Since 2010, the advent of direct oral anticoagulants (DOACs) has reshaped the clinical landscape of anticoagulation therapy. This class mainly includes apixaban, rivaroxaban, edoxaban and dabigatran.
Among them, apixaban (BMS/Pfizer) and rivaroxaban (Johnson & Johnson/Bayer) are core global anticoagulant products. With fast onset, few drug interactions, no need for frequent monitoring and other advantages, they have completely redefined the anticoagulation field. In 2025, global sales of apixaban reached $22.4 billion. Even so, all existing anticoagulants share an insurmountable inherent contradiction: while blocking pathological thrombus formation, they inevitably interfere with normal physiological hemostasis, leading to serious adverse reactions such as intracranial hemorrhage and gastrointestinal bleeding — the biggest constraint in clinical anticoagulation therapy.
The emergence of factor XI (FXI) inhibitors provides a revolutionary solution to this dilemma.FXI is a plasma serine protease, mainly synthesized by hepatocytes and circulating as a zymogen. It is a key component of the intrinsic coagulation pathway, triggering the formation of thrombin and fibrin primarily through a series of contact activation reactions.
Studies have confirmed that FXI is essential for pathological thrombus formation but, in most cases, not required for normal hemostasis.
FXI inhibitors are therefore expected to uncouple thrombus formation from hemostatic function, offering a safer alternative for anticoagulation therapy.
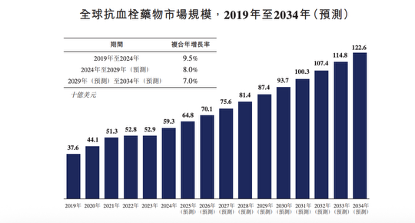
Meanwhile, the continuous expansion of the market underscores the urgent demand for safe and highly effective new drugs. According to Frost & Sullivan, the global antithrombotic drug market grew from $37.6 billion in 2019 to $59.3 billion in 2024, and is projected to reach $87.4 billion by 2029 and $122.6 billion by 2034.Behind this massive market lies the pursuit of “safer anticoagulation”.
 Picture source: Prospectus from Suzhou Ribo Life Science Co., Ltd.
Picture source: Prospectus from Suzhou Ribo Life Science Co., Ltd.
02 Global Race
According to Yaozh Data, there are more than 30 active anticoagulant drugs in global R&D, among which over 20 are FXI/FXIa inhibitors, accounting for more than 60%, making it one of the core tracks for new anticoagulant R&D. No FXI/FXIa inhibitor has been approved globally to date. Novartis’ abelacimab, Bayer’s asundexian, and Johnson & Johnson’s milvexian have all advanced to Phase III clinical trials, and the global race has entered an intensified, white-hot phase.
Monoclonal antibody (mAb) FXI inhibitors are administered subcutaneously or intravenously with an extremely long half-life, enabling dosing intervals of one month or longer, which greatly improves patient compliance.
The star product in this field is Novartis’ abelacimab. In February 2025, Novartis acquired Anthos Therapeutics for up to $3.075 billion, with the core asset being abelacimab, an anti-FXI mAb. Abelacimab is a novel, highly selective fully human monoclonal antibody that binds tightly to coagulation factor XI and prevents its activation, thereby inhibiting both factor XI and its activated form, factor XIa, to prevent thrombosis.
Phase II clinical data showed that, in patients with atrial fibrillation (AF), abelacimab significantly reduced bleeding events across all primary and secondary endpoints compared with standard DOAC therapy. Once-monthly subcutaneous injection of 150 mg abelacimab reduced FXI levels by 99%, with durable efficacy exceeding 2 years.
Multiple Phase III trials of abelacimab are currently underway, with the primary indication being the prevention of stroke and systemic embolism in patients with atrial fibrillation. Data are expected to be released in 2026.
Atrial fibrillation is one of the most common sustained cardiac arrhythmias, affecting an estimated 500 million people worldwide, and the associated stroke risk is five times higher than in those without the condition. Anticoagulant therapy for the prevention of cardioembolic stroke is therefore a key focus in the treatment of atrial fibrillation.
Regeneron has developed two differentiated monoclonal antibodies: REGN9933 targets the A2 domain of FXI, blocking the activation of FXI by FXIIa (without affecting FIIa activation), mimicking the FXII-deficient state and suitable for patients at high bleeding risk; REGN7508 targets the catalytic domain, providing potent inhibition of FXI-mediated coagulation, and is being developed across various anticoagulant indications, especially for patients requiring robust anticoagulant efficacy, such as those undergoing joint replacement or with cancer-associated venous thromboembolism (VTE).
In a Phase II trial after knee replacement, the primary clinical endpoint was the incidence of VTE up to 12 days post-surgery. Data showed that the VTE rate for REGN7508 (7.1%) was superior to enoxaparin (20.6%) and numerically lower than apixaban (12.4%); REGN9933 was numerically lower than enoxaparin (17.2% vs 20.6%). No major bleeding events occurred in either group.
Oral FXI inhibitors offer convenient administration and clear manufacturing advantages, making them well-suited for long-term anticoagulation settings such as atrial fibrillation and stroke prevention, representing one of the competitive directions of the future market.
In this track, Bayer’s asundexian has achieved a landmark breakthrough. Asundexian is one of the world’s most advanced oral FXIa inhibitors. Although its Phase III trial for atrial fibrillation was terminated early due to insufficient efficacy, its Phase III secondary stroke prevention study OCEANIC-STROKE was a resounding success. OCEANIC-STROKE was a Phase III trial comparing asundexian plus antiplatelet therapy head-to-head against placebo plus antiplatelet therapy for secondary prevention in stroke patients.
In February 2026, Bayer announced positive results from the OCEANIC-STROKE study of asundexian. In patients with non-cardioembolic ischemic stroke or high-risk transient ischemic attack (TIA), asundexian added to standard antiplatelet therapy significantly reduced the risk of recurrent ischemic stroke by 26% without increasing the risk of major bleeding as defined by the International Society on Thrombosis and Haemostasis (ISTH). This is the world’s first successful Phase III study of an FXIa inhibitor, validating the druggability of this target.
Milvexian is also a highly selective oral FXIa inhibitor, co-developed by Bristol-Myers Squibb (BMS) and Johnson & Johnson, and has now entered Phase III clinical trials.Its Phase III trial in acute coronary syndrome (ACS) was terminated early for failing to meet the primary efficacy endpoint, and development is now fully focused on stroke prevention.
03Domestic Innovation Shines Brightly
Amid the global wave of FXI/FXIa inhibitors, Chinese innovative pharmaceutical companies have not been left behind. Instead, they have achieved breakthroughs in two cutting-edge directions — siRNA and monoclonal antibodies (mAbs), demonstrating R&D capabilities comparable to international peers.
Unlike small-molecule drugs that typically require daily administration, FXI-targeted siRNA therapeutics feature long-acting efficacy, enabling extended dosing intervals and sustained reduction of FXI protein and activity, thereby improving patient compliance and lowering overall treatment costs.In addition, compared with antibodies, the synthetic nature and liver-specific delivery of siRNA drugs result in lower immunogenicity and fewer drug-resistant antibodies, whereas protein-based antibody drugs carry an inherently higher risk of immunogenicity.
With the advantages of ultra-long-acting efficacy and low immunogenicity, siRNA therapeutics have emerged as a highly promising new direction in the field of FXI/FXIa inhibitors. Such therapies can be administered as infrequently as once per quarter or even once every six months, providing an innovative solution to the long-standing problem of poor compliance in long-term anticoagulation therapy. In this field, Chinese innovative pharma companies are at the global forefront.
RBD4059 from Ribo Life Science (Suzhou) Co., Ltd. (Ribolia) is the world’s first siRNA drug for thrombosis to enter clinical development, and is currently in a Phase II trial for stable coronary artery disease (SCAD).In Phase I studies, RBD4059 demonstrated potent efficacy: the mean maximum percentage changes from baseline in FXI activity in the 50 mg, 150 mg, 400 mg and 600 mg cohorts were 67.5%, 81.0%, 85.8% and 91.6%, respectively, with durable efficacy observed up to Day 169 at the study endpoint.
RBD4059 combines the advantages of FXI targeting with siRNA drug technology, delivering strong efficacy alongside a notable safety profile.
Clinical and preclinical data show that RBD4059 can greatly reduce the bleeding risk associated with traditional anticoagulants. Furthermore, the long-acting nature of siRNA therapy may significantly improve patient compliance, positioning RBD4059 as a potential optimal treatment option for a broad population of patients with thrombotic diseases.
SRSD107 from Jingyin Pharmaceuticals is another FXI/FXIa siRNA drug, currently in a Phase II trial for VTE prevention after knee replacement. In May 2025, Jingyin Pharmaceuticals announced a major collaboration with CRISPR Therapeutics, receiving a $95 million upfront payment and over $800 million in milestone payments for the joint development of SRSD107.
Hengrui Medicine is a leader among domestic developers of FXI/FXIa inhibitors. SHR-2004 is a humanized monoclonal antibody targeting FXI, independently discovered by Hengrui Medicine, which exerts anticoagulant effects by blocking the cascade of the intrinsic coagulation pathway.Phase II clinical data for VTE prevention in patients with ovarian cancer, presented at ESGO 2025, showed that the VTE rate in the SHR-2004 group (12.6%) was significantly lower than that in the active control group (20.2%), with no major VTE or related deaths and favorable safety and tolerability.
04 Conclusion
From the century-old dilemma of “anticoagulation inevitably leads to bleeding” to the dawn of “safe anticoagulation”, FXI/FXIa inhibitors represent a fundamental paradigm shift in the treatment of thrombotic diseases. Although challenges remain, the positive Phase III results for asundexian and Novartis’ high-value acquisition of Anthos Therapeutics clearly signal one thing: the FXI/FXIa inhibitor race has ignited, and the wave of innovation is irreversible.
By editorRead more on
- Boehringer Ingelheim’s new oral drug “Zonaitinib” has been approved for a new indication in China May 22, 2026
- Class 1 innovative drug Fazanleisen tablets approved May 22, 2026
- Shijiazhuang Pharmaceutical Group’s budesonide enteric-coated capsules approved! This poses a challenge to generic drug manufacturers, weakening the original drug’s competitive advantage. May 22, 2026
- Two world-first drugs from Chipscreen Biosciences have been launched in the Macau market, further improving patient accessibility! May 22, 2026
- Beimei Prostol Eye Drops Receives Drug Registration Certificate May 22, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



