-
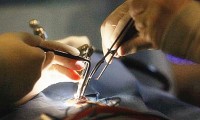
New tech “lights up” nerves to identify, reduce accidental damage
- Source: massdevice
- 940
- August 21, 2017
-
NeoSync secures $13m in financing with Valiance Life Science Investments and current Investors
- Source: patientmonitoring
- 1,075
- August 17, 2017
-
GSK opens new aluminium salts facility in Scotland
- Source: news.open-medical
- 1,049
- August 17, 2017
-
MC10 and Shirley Ryan AbilityLab to clinically study BioStampRC System
- Source: massdevice
- 1,068
- August 16, 2017
-
Atlas awarded £2m grant for infectious disease test
- Source: news.open-medical
- 786
- August 16, 2017
-
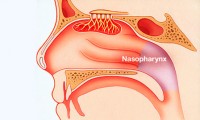
New CUHK study shows early nasopharyngeal cancer detection with EBV DNA
- Source: medicaldevice-network
- 886
- August 15, 2017
-
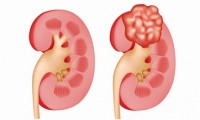
NICE turns down kidney cancer drugs
- Source: pharmatimes
- 845
- August 15, 2017
-
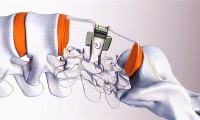
FDA approves Renovis Surgical’s 3D-printed posterior lumbar interbody fusion systems
- Source: orthopedicdevices
- 731
- August 15, 2017
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.