Hengrui Medicine launches two major initiatives! Its HER3 ADC leader SHR-A2009 receives IND approval, and its adebenone enters the KRAS-mutant solid tumor market.
May 14, 2026
Source: drugdu
 62
62
 On May 13, 2026, the latest announcement on the official website of the Center for Drug Evaluation (CDE) of the National Medical Products Administration showed that two subsidiaries of Hengrui Medicine simultaneously obtained acceptance qualifications for two Investigational New Drug (IND) applications: Suzhou Shengdiya Biopharmaceutical's independently developed SHR-A2009 for injection (a Class 1 therapeutic biological product) and Shanghai Shengdi Pharmaceutical's Adebelimab injection (a Class 2.2 therapeutic biological product), with acceptance numbers CXSL2600515 and CXSL2600514 respectively, both accepted on the same day. This is another impressive achievement for Hengrui Medicine in the field of innovative drugs this month, following the acceptance of four biological drugs in a single day on April 17, demonstrating its strong R&D and translation capabilities.
On May 13, 2026, the latest announcement on the official website of the Center for Drug Evaluation (CDE) of the National Medical Products Administration showed that two subsidiaries of Hengrui Medicine simultaneously obtained acceptance qualifications for two Investigational New Drug (IND) applications: Suzhou Shengdiya Biopharmaceutical's independently developed SHR-A2009 for injection (a Class 1 therapeutic biological product) and Shanghai Shengdi Pharmaceutical's Adebelimab injection (a Class 2.2 therapeutic biological product), with acceptance numbers CXSL2600515 and CXSL2600514 respectively, both accepted on the same day. This is another impressive achievement for Hengrui Medicine in the field of innovative drugs this month, following the acceptance of four biological drugs in a single day on April 17, demonstrating its strong R&D and translation capabilities.
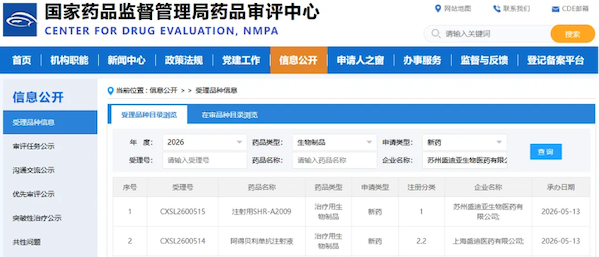 SHR-A2009: A leading global HER3 ADC, targeting the pain points in the diagnosis and treatment of EGFR-resistant lung cancer.
SHR-A2009: A leading global HER3 ADC, targeting the pain points in the diagnosis and treatment of EGFR-resistant lung cancer.
The SHR-A2009 injectable drug , which has been accepted for approval , is a new generation HER3-targeting antibody-drug conjugate (ADC) independently developed by Hengrui Medicine, and has now joined the first tier of global HER3 ADC research and development. This product employs precise molecular design, consisting of a fully human HER3 IgG1 monoclonal antibody, an enzyme-cleavable linker, and a topoisomerase I (Top I) inhibitor payload. With an average drug-to-antibody ratio (DAR) of 4, it can precisely target HER3-positive tumor cells, efficiently delivering the cytotoxic payload to the lesion, achieving specific killing of tumor cells while balancing efficacy and safety.
HER3, a core member of the EGFR family, is highly expressed in over 80% of non-small cell lung cancer (NSCLC) and is one of the core causes of resistance to EGFR-TKI targeted therapy. For a long time, there has been a lack of effective targeted treatment options, representing a significant unmet medical need. Leveraging its unique molecular design advantages, SHR-A2009 has demonstrated excellent anti-tumor activity and good safety in early clinical studies. Currently, the product has initiated its first international multicenter Phase III clinical trial (SHR-A2009-301), a head-to-head comparison with platinum-based chemotherapy, for the treatment of EGFR-mutant advanced NSCLC that has failed EGFR-TKI therapy. It is expected to receive Fast Track designation from the US FDA in 2025, potentially becoming one of the world's first approved HER3 ADC drugs, filling a gap in related therapeutic areas.
This IND application marks another key milestone in the clinical development of SHR-A2009. Industry experts speculate that this product will further expand its application scenarios, explore its therapeutic potential in other HER3-highly-expressing solid tumors, and advance combination therapy regimens with immunotherapy and targeted therapy, continuously expanding the potential beneficiary population.
Adebenone: Continuously expanding its applications since its market launch, combining with KRAS inhibitors to tackle refractory tumors.
On the same day , adebenonemab injection (trade name: Arelei®), an anti-PD-L1 monoclonal antibody independently developed by Hengrui Medicine, was also accepted for approval . It is the first domestically produced PD-L1 inhibitor approved in China for first-line treatment of extensive-stage small cell lung cancer (ES-SCLC). To date, adebenonemab has been successfully approved in China for two core indications: first-line treatment of extensive-stage small cell lung cancer in combination with chemotherapy, and first-line treatment of unresectable locally advanced or metastatic squamous non-small cell lung cancer (sqNSCLC) in combination with chemotherapy, significantly improving the survival prognosis of Chinese lung cancer patients and providing a better option for clinical treatment.
This Class 2.2 new drug clinical trial application is an important step in expanding the indications for adebelimumab after its market launch. According to an announcement released by Hengrui Medicine on May 8, 2026, this clinical trial will focus on exploring the combination of adebelimumab and HRS-6093 (a novel oral KRAS G12D inhibitor) for the treatment of advanced malignant solid tumors with KRAS G12D mutations. It is understood that KRAS G12D is the most common subtype of KRAS mutations, accounting for approximately 30% of all KRAS mutations, and has long been considered "undruggable," highlighting the urgent clinical need for its treatment.
The combination therapy of adebenone and HRS-6093 represents a new approach to precision medicine with "targeted + immune" synergistic treatment. It is expected to break the treatment dilemma of KRAS G12D mutant solid tumors and bring new hope to patients with refractory tumors such as pancreatic cancer, colorectal cancer, and non-small cell lung cancer.
Accelerating pipeline gains, Hengrui builds a comprehensive tumor treatment system combining ADC and immunotherapy.
The simultaneous acceptance of two core products by the CDE (Center for Drug Evaluation) is a testament to Hengrui Medicine's high-intensity R&D investment and continuous efforts in innovation transformation. Since 2026, Hengrui Medicine has witnessed a continuous surge in innovative achievements: the world's first anti-PD-L1/TGF-βRII bispecific antibody fusion protein, Rellavpoxetine α injection, has been successfully approved for marketing; eight new drug applications have been accepted; and multiple core products are steadily progressing to key clinical stages. The innovation pipeline is showing a positive trend of "multiple breakthroughs and continuous gains."
In the ADC field, Hengrui Medicine has built a complete pipeline covering core oncology targets such as HER2, HER3, and c-Met, forming a differentiated competitive advantage. On April 17, 2026 alone, clinical trial applications for three ADC products (SHR-A1811, SHR-A2009, and SHR-1826) were simultaneously accepted by the CDE, marking the official entry of Hengrui Medicine's full-industry chain layout in the ADC field into an accelerated harvest period.
In the field of immunotherapy, adebenone, as Hengrui Medicine's core immunotherapy product, is comprehensively advancing the expansion of indications and the exploration of combination therapies, covering multiple mainstream tumor types such as lung cancer, liver cancer, gastric cancer, and esophageal cancer. It forms a strong synergistic effect with the company's targeted drugs and ADC products, and gradually builds a comprehensive and multi-level tumor treatment system.
With the orderly progress of subsequent clinical trials, Hengrui Medicine is expected to continue to launch more high-quality innovative treatment products, benefiting cancer patients in China and even the world, further consolidating its leading position in the domestic innovative drug field, and promoting the high-quality development of China's pharmaceutical industry towards innovation and internationalization.
https://mp.weixin.qq.com/s/XWoTXyVZY-5hM9gUAvfRyQ
By editorRead more on
- First Generic Just Approved, Third Filing for Market Authorization! May 15, 2026
- A $15.2 billion deal, the top player blows the show! May 15, 2026
- //news.yaozh.com/archive/48019.html Abesilli tablets May 15, 2026
- Gilead’s new HIV drug “exceeds expectations” May 15, 2026
- Lispro insulin receives EU approval, marking the first time domestically produced insulin has broken the European monopoly on original drugs. May 15, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



