-
A Scientifically-Supported App for Early Stage Dementia by SMART Brain Ageing
- Source: HitConsultant
- 1,301
- July 10, 2018
-
Biotech Crinetics Therapeutics Raises $86 million in IPO
- Source: FierceBiotech
- 1,236
- June 26, 2018
-
Nanobiotix NBTXR3 in Late Phase Clinical Trial
- Source: FierceBiotech
- 989
- June 26, 2018
-
Re-Investigation on FDA Approved Anti-Gout Drug Uloric
- Source: FiercePharma
- 1,021
- June 25, 2018
-
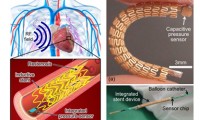
Smart Stent to Continuously Monitor Blood Flow
- Source: ScienceDaily
- 2,017
- June 22, 2018
-
Gout Treatment – Shift from Phase 2 to Phase 3 Trial
- Source: FierceBiotech
- 957
- June 21, 2018
-
Imutex Universal FLU Vaccine Reaching Phase 3
- Source: FiercePharma
- 1,334
- June 21, 2018
-
What is COPD and how is it Treated?
- Source: HealthLine
- 1,169
- June 19, 2018
-
Antidepressants or Talk Therapy? – Know with EEG
- Source: ScienceDaily
- 964
- June 19, 2018
-
Calliditas Therapeutics Files to Raise $75 M for Kidney Disease Trial
- Source: Fierce Biotech
- 1,167
- June 15, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.