-
BioNTech and Duality Biologics add third ADC candidate to oncology partnership
- Source: drugdu
- 72
- August 11, 2023
-
MSD and Astex announce expansion to oncology drug discovery collaboration
- Source: drugdu
- 74
- August 10, 2023
-
Alkermes, Nearing Oncology Split, Posts Big Q2 Sales Gain After Winning J&J Arbitration Case
- Source: drugdu
- 71
- July 28, 2023
-
J&J Exceeds Q2 Expectations, Oncology and Immunology Top Pharma Drivers
- Source: drugdu
- 65
- July 24, 2023
-
Bayer needs ‘midsize acquisition’ to reach $10B oncology sales goal, exec says
- Source: drugdu
- 77
- June 15, 2023
-
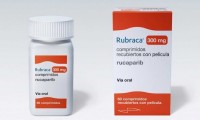
After selling Rubraca, Clovis Oncology unveils CRL for potential label expansion
- Source: drugdu
- 114
- June 7, 2023
-
Quest Expands in Cancer Testing With $300M Haystack Oncology Acquisition
- Source: drugdu
- 131
- April 29, 2023
-
BullFrog AI , Sage Group partner to develop oncology assets
- Source: drugdu
- 79
- April 25, 2023
-
Caris Life Sciences and Flare to collaborate for oncology pipeline advancement
- Source: drugdu
- 131
- April 17, 2023
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.