FDA Approves Pulmonx’s Zephyr Device to Treat Emphysema
July 4, 2018
Source: FierceBiotech
 1,203
1,203
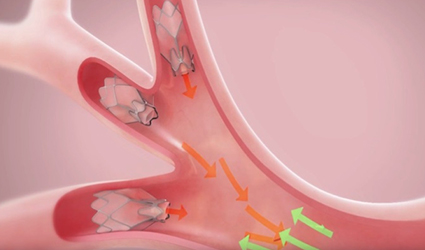
The premarket approval of Pulmonx’s Zephyr Endobronchial Valve to treat severe emphysema has green-lighted by the FDA. Zephyr is a pencil eraser-sized device delivered directly to the lung in a minimally invasive procedure. After the successful delivery, the device stops air from entering diseased parts of the lung during breathing. This mechanism prevents the air from getting trapped by damaged alveoli and stops the breathing difficulties.
Pulmonx tested the Endobronchial Valve in a clinical trial, which consisted of 190 patients affected with lung disease. Around two-thirds of the participants received a combined therapy including Zephyr and standard medical management, such as pulmonary rehabilitation and medicines. The remaining participants received only standard medical management. After a year, nearly 48% of the patients had experienced greater improvement in pulmonary function capacity, compared to 17% of patients who experienced only medical improvement. This remarkable data led to the approval by FDA.
The Endobronchial Valve acts as an alternative to surgical procedures such as lung volume reduction and lung transplants for the emphysema patients.
Tina Kiang from FDA stated, “Treatment options are limited for people with emphysema who have severe symptoms that have not improved from taking medicines; This novel device is a less invasive treatment that expands the options available to patients.”
By DduRead more on
- AZ’s Farxiga Gets FDA Priority Review For Heart Failure January 8, 2020
- Smartphone Based Diagnosis to Identify Mosquitoes Transmitting Infection September 5, 2018
- Global Recall of CyPass Micro-Stent by Alcon September 3, 2018
- 3D Human Liver Implant – The Need of the Hour August 29, 2018
- IACS – An Advanced Cell Sorter August 29, 2018
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



