Non-profit receives FDA approval to supply cheaper OTC naloxone
August 2, 2023
Source: drugdu
 376
376
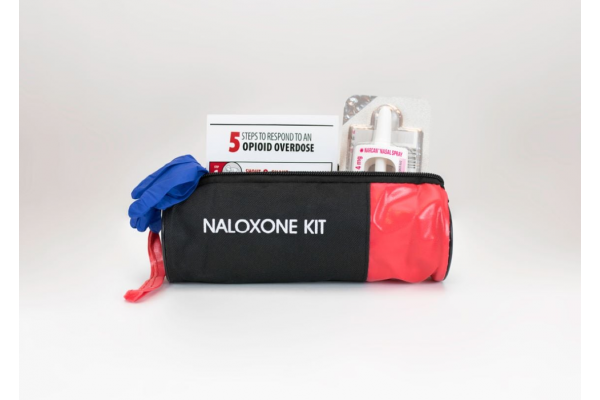
The US Food and Drug Administration (FDA) approved the second over-the-counter (OTC) naloxone nasal spray product, RiVive (3mg) by non-profit pharmaceutical company Harm Reduction Therapeutics, for the emergency treatment of known or suspected opioid overdose.
This approval follows the agency’s approval of Emergent BioSolutions’ Narcan in March 2023, as the agency has recently increased efforts to address the opioid crisis.
The FDA also approved the first generic non-prescription naloxone drug in July 2023. Naloxone reverses opioid overdoses by blocking opioid receptors, thus preventing the drugs’ addictive effects. Harm Reduction Therapeutics has announced that the drug will be available by early 2024 and be primarily supplied to state governments and US harm organisations at “costs lower than other opioid antagonist nasal sprays”.
The US-based non-profit organisation has said that the first 200,000 doses (10% of projected initial annual product production) of RiVive will be available free of charge. Side effects of RiVive include diarrhoea, fever, body aches and more.
In a press release, Harm Reduction Therapeutics announced that no company, entity or individual will receive profit from RiVive’s sales. The non-profit encouraged funding partners to direct any financial support to reduce the drug’s price tag, improve manufacturing capacity, and thus improve the drug’s accessibility. This follows Emergent Biosolutions’ April 2023 announcement that Narcan will also be available at a lower price for similar reasons.
During the 12-month period ending in February 2023, more than 105,000 reported fatal overdoses occurred with a primary cause related to synthetic opioids like illicit fentanyl, as per the FDA. The approval of OTC naloxone is one of many actions the agency has taken to address the rising opioid problem.
This year, the FDA has also targeted the crisis by expanding options for opioid disposal through the implementation of a mail-back envelope system, and the progression of its overdose prevention framework.
In a press release, FDA commissioner Dr Robert Califf said: “The agency has long prioritised access to naloxone products, and we welcome manufacturers of other naloxone products to discuss potential nonprescription development programs with the FDA.”
Harm Reduction Therapeutics is developing RiVive under a commercial supply agreement with US-based Catalent. In this deal, Catalent will manufacture the therapy at its 180,000ft² inhalation development facility in Morrisville, North Carolina.
By editor
Read more on
- Jingfang Pharmaceuticals’ small molecule drug receives Fast Track designation from the US FDA for lung cancer June 8, 2026
- Once a month! The world’s first new drug for IgA nephropathy has been approved for marketing in China. June 8, 2026
- The world’s first Class 1 new drug from a Guangdong pharmaceutical company has been approved for marketing June 8, 2026
- GenSci Pharmaceutical’s self-developed Class 1 new drug has been approved for clinical trials for two indications, covering pediatrics and neurology. June 8, 2026
- Yunnan Baiyao Group’s Central Research Institute’s INB301 injection IND has received FDA clinical trial approval, achieving a milestone breakthrough with “dual applications in China and the United States” June 8, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



