Heavy weight! Two products have been recognized as “Innovative Medical Devices”
March 24, 2025
Source: drugdu
 346
346
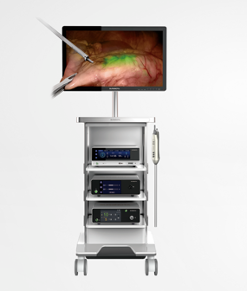 According to the China Medical Device Innovation and Priority Approval Database, two products are proposed to be approved as innovative medical devices in Beijing in recent public announcements released by local drug supervision and administration bureaus across the country. They are: the 3D4K fluorescence electronic chest and abdominal cavity endoscope from Seno Micro Medical Technology (Beijing) Co., Ltd. (hereinafter referred to as Seno Micro), and the 3D4K fluorescence endoscope image processor.
According to the China Medical Device Innovation and Priority Approval Database, two products are proposed to be approved as innovative medical devices in Beijing in recent public announcements released by local drug supervision and administration bureaus across the country. They are: the 3D4K fluorescence electronic chest and abdominal cavity endoscope from Seno Micro Medical Technology (Beijing) Co., Ltd. (hereinafter referred to as Seno Micro), and the 3D4K fluorescence endoscope image processor.
Multiple innovative achievements have been implemented
Founded in 2015, Sinovac has been dedicated to the fields of minimally invasive surgery and minimally invasive interventional therapy for tumors, striving to become the world's preferred provider of comprehensive minimally invasive surgical solutions. After years of development, Sino Micro has built four major platforms: surgical energy, intelligent anastomosis, surgical vision, and tumor ablation, providing a comprehensive solution for minimally invasive surgical treatment worldwide.
At present, Sainuo Micro's product pipeline mainly includes ultrasonic soft tissue cutting hemostasis equipment, ultrasonic high-frequency surgical integrated surgical equipment, ultrasonic high-frequency large vessel closure integrated surgical equipment, electric endoscope linear cutting stapler, 3D full HD, 3D full HD fluorescence, 3D4K, 3D4K fluorescence photoelectron thoracoscopy and laparoscopy, as well as 4K and 4K fluorescence optical hard mirror imaging systems, white light and fluorescence light sources, pneumoperitoneum machines, steep pulse therapy devices, microwave ablation devices, radiofrequency ablation devices, etc. Its product pipeline covers equipment and instruments throughout the entire minimally invasive surgery process, with multiple products having obtained CE certification and approval from NMPA and FDA.
DeepEye ™ 3D4K fluorescence electronic chest laparoscope is a product under the vision platform of Sino Microsurgery, which was certified and launched in July 2024 (Zhejiang Medical Equipment Registration: 20242061623). It is reported that this product combines 3D, 4K technology with fluorescence imaging technology, which can provide better depth perception, clearer images, richer details, and more accurate target positioning.
Based on the combination of multiple technologies, Sainuo Micro's 3D4K fluorescence electronic chest laparoscope combines excellent 3D performance, 4K performance, fluorescence performance, and algorithm performance, making the operation of the product more convenient. It can support multiple departments, multiple surgical procedures, and multi-user parameter settings and saving.
Among them, the 3D performance of the product is applicable to the original DeepEye ™ 3D precise spatiotemporal matching technology, and its integrated electronic mirror design, without focusing, eliminates the problem of optical mirror 3D effect mismatch; In terms of 4K performance, from the camera, processor to the display, it supports full link 4K resolution ultra high definition output, which can finely display fine blood vessels, small nerves, and fascia layers, and clearly distinguish tissue boundaries; DeepEye is applied in the fluorescence performance ™ NIR/ICG fluorescence imaging technology, which is a top level fluorescence high sensitivity technology in the industry, can highlight details and contours such as lesions, nerves, and blood vessels; In terms of algorithm performance, it also combines intelligent noise reduction, edge enhancement, vascular enhancement, and intelligent supplementary lighting functions
In addition to this product, Sino Micro has also achieved multiple results in recent years. In October 2024, Dophi ® The N3000 steep pulse therapy device has been approved for market by the US Food and Drug Administration (FDA). This product is also the first irreversible electroporation device in China to receive both CE and FDA certifications. With the passage of time and the continuous R&D investment of Sinovac, more innovative products will be launched in the future, which is worth looking forward to.
The gradual rise of domestic substitution forces
Medical endoscope is a commonly used medical device in modern medicine. In terms of categories, there are a wide variety of endoscopes, including gastroscopy, colonoscopy, laryngoscopy, esophagoscopy, laparoscopy, thoracoscopy, cholangioscopy, cystoscopy, ureteroscopy, nephroscopy, hysteroscopy, vascular endoscopy, arthroscopy, etc.
With the advancement of technology, medical endoscopes are gradually being combined with 4K, 3D, disposable technology, special light (such as fluorescence) imaging technology, ultra-fine medical endoscope technology, big data, artificial intelligence and other technologies, and the market share of medical endoscopes is also constantly increasing.
Based on the continuous increase in market share, medical endoscope products have also ushered in an explosive period in recent years.
According to the data from the China Medical Device Screening System, more than 2886 endoscope series products have been approved for market in China, and the registration trend of such products is also increasing year by year. By 2024, the registration number of such products will reach the highest value in nearly five years.
Among the approved products, endoscopic imaging systems have the highest number, followed by endoscopic image processors. However, by 2024, the number of registered endoscopic image processors has exceeded that of endoscopic imaging systems.
For a long time, the domestic endoscope market in China has been firmly controlled by foreign brands such as Olympus, Pentax, and KARL STORS, which once held about 90% of the market share in China. Since 2020, this phenomenon has undergone some changes, and domestic equipment companies in the field of endoscopes have sprung up like mushrooms after rain. At present, among the approved endoscopes in China, domestic products far exceed imported products, accounting for 82.85% of the total.
According to data, the total market share of overseas companies in the Chinese endoscope market has dropped to about 60%, while domestic brands such as Aohua Endoscope, Mindray Medical, Kaikai Medical, and Opumand have entered the top ten in terms of sales, accounting for about 15% of the market share.
Conclusion
With the support of policies and the promotion of continuous technological development, the "gold content" of domestic medical devices is becoming increasingly high, and local medical devices are continuously shining with new brilliance.
Source: https://news.yaozh.com/archive/45168.html
By editorRead more on
- First Generic Just Approved, Third Filing for Market Authorization! May 15, 2026
- A $15.2 billion deal, the top player blows the show! May 15, 2026
- //news.yaozh.com/archive/48019.html Abesilli tablets May 15, 2026
- Gilead’s new HIV drug “exceeds expectations” May 15, 2026
- Lispro insulin receives EU approval, marking the first time domestically produced insulin has broken the European monopoly on original drugs. May 15, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



