Domestic neural device giant sprints for IPO
September 7, 2024
Source: drugdu
 416
416
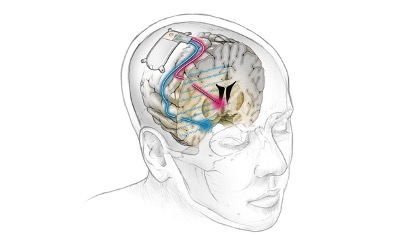 Recently, Beijing Pinchi Medical Equipment Co., Ltd. (hereinafter referred to as "Pinchi Medical") completed its IPO counseling and registration with the Beijing Securities Regulatory Bureau, with the counseling agency being China International Capital Corporation (CICC).
Recently, Beijing Pinchi Medical Equipment Co., Ltd. (hereinafter referred to as "Pinchi Medical") completed its IPO counseling and registration with the Beijing Securities Regulatory Bureau, with the counseling agency being China International Capital Corporation (CICC).
According to the coaching work arrangement, CICC plans to complete the coaching plan for Pinchi Medical in November December this year and apply for coaching acceptance from the Beijing Securities Regulatory Bureau. After completing the coaching acceptance, Pinchi Medical may apply for IPO.
This is another brain pacemaker company planning to go public in 2023, following Jingyu Medical's IPO guidance filing. According to reports from Smart Medical Devices, the only two local companies that mass produce brain pacemakers have now embarked on the path of IPO and planned to go public.
Tsinghua endorsement, Pinchi Medical is aiming for IPO
Pinchi Medical was founded in 2008 and is a high-tech enterprise specializing in the research, development, production, and sales of a series of neural regulation products such as brain pacemakers, vagus nerve stimulators, spinal cord stimulators, and sacral nerve stimulators. It is committed to providing advanced treatment methods for patients with functional neurological diseases such as Parkinson's disease, epilepsy, pain, and urinary incontinence.
Tracing back to Pinchi Medical, it can be found that its foundation is very profound. Although it was founded in 2008, in fact, as early as around the millennium, its founding team has been engaged in tackling key technologies and products of implantable neural regulation such as brain pacemakers. There is also a key figure in the team - Li Luming, an aerospace research expert of Tsinghua University. He was elected as an academician of the CAS Member in November 2023, and served as the vice secretary of the Party Committee and president of Tsinghua University.
Pinchi Medical has also formed an inseparable bond with Tsinghua University. Li Luming once served as the chief scientist of Pinchi Medical, and the general manager of Pinchi Medical, Hao Hongwei, is a researcher and doctoral supervisor at Tsinghua University. He is also the deputy director of the National Engineering Laboratory for Neuromodulation Technology and the deputy director of the Institute of Human Machine and Environmental Engineering. He has a deep understanding and contribution to the operation management and technological development of Pinchi Medical. And the chief engineer of Pinchi Medical, Wen Xiongwei, is also a high-precision and cutting-edge talent from Tsinghua University. He once served as the director of the Laboratory of Human Machine Environment and Medical Engineering at the School of Aerospace Science and Technology, Tsinghua University, and played an important role in promoting Pinchi Medical's technological research and product innovation.
At present, Pinchi Medical has achieved numerous research results with completely independent intellectual property rights, obtained registration certificates for a series of brain pacemakers, vagus nerve stimulators, sacral nerve stimulators, and spinal nerve stimulators, and launched them for sale, breaking the monopoly of American companies in the field of neural regulation products.
According to data from Yaozhi Medical Equipment, nearly 20 products from Pinchi Medical have entered the innovation approval channel, including spinal nerve stimulation test electrodes, deep brain electrical stimulation systems, etc.
On May 28, 2024, Pinchi Medical approved four innovative products - the implantable deep brain electrical stimulation electrode lead kit, the dual channel rechargeable implantable deep brain electrical stimulation pulse generator kit, the dual channel implantable deep brain electrical stimulation pulse generator kit, and the implantable deep brain electrical stimulation extension lead kit. They are the first directional deep brain electrical stimulation products in China, which can stimulate the subthalamic nucleus or medial pallidum through complementary use, and are used for combination therapy of late stage levodopa responsive Parkinson's disease patients who cannot effectively control certain symptoms with drugs. It is also highly anticipated whether Pinchi Medical can be successfully launched this time.
The market is vast, and domestic overtaking and acceleration are underway
Brain pacemaker implantation is commonly known as Deep Brain Stimulation (DBS). When it comes to brain pacemakers, the general public may be relatively unfamiliar with them. However, implantable neural control products such as brain pacemakers are "electronic drugs" for patients with functional neurological diseases such as Parkinson's disease, epilepsy, pain, and urinary incontinence, which can improve their quality of life.
According to the latest research report released by Grand View Research, the global market size of DBS in 2016 was approximately 796 million US dollars, with a predicted compound annual growth rate of 11.5% for the DBS market during the forecast period. By 2025, the global DBS market capacity is expected to exceed $2.1 billion.
In China, functional neurosurgery diseases have a large number of patients. With the deepening of the aging of the population in China, the number of patients with Parkinson's disease is conservatively estimated to be about 2.5 million nationwide, but less than 1% receive neuromodulation therapy.
As an active implantable high-end medical device, although brain pacemakers have obvious therapeutic effects, the development difficulty is extremely high. For a long time, the domestic brain pacemaker market has been monopolized by foreign brands such as Medtronic, Poco, Abbott, etc., and high pricing has deterred many patients.
In recent years, domestic substitution forces have emerged, with Jingyu Medical and Pinchi Medical creating multiple domestic firsts, and domestic overtaking is accelerating. In addition to Pinchi Medical, which is rushing for IPO this time, Jingyu Medical is also a rising star in the domestic brain pacemaker industry. Jingyu Medical is an innovative high-tech medical device company that integrates research and development, production, and sales of deep brain electrical stimulation systems. It has complete independent intellectual property rights and continuously develops new technologies and products.
In 2014, Jingyu Medical was included in the special approval channel for innovative medical devices by the national drug regulatory authorities; In 2015, its dual channel implantable deep brain electrical stimulation pulse generator kit, implantable deep brain electrical stimulation electrode lead kit, and implantable deep brain electrical stimulation extension lead kit were approved for sale. In 2017, Jingyu Medical's third-generation product - the brain pacemaker for treating drug addiction - was included in the special approval channel for innovative medical devices by the national drug regulatory department.
In April 2022, its dual target deep brain electrical stimulation technology and products for the treatment of drug addiction were granted breakthrough medical device qualification by the US Food and Drug Administration (FDA). This is also the first time that Chinese companies have obtained breakthrough medical device qualifications in the field of neurological and psychiatric disorders. Currently, Jingyu Medical is further expanding in the field of mental illness.
Source: https://news.yaozh.com/archive/44160.html
By editorRead more on
- Jingfang Pharmaceuticals’ small molecule drug receives Fast Track designation from the US FDA for lung cancer June 8, 2026
- Once a month! The world’s first new drug for IgA nephropathy has been approved for marketing in China. June 8, 2026
- The world’s first Class 1 new drug from a Guangdong pharmaceutical company has been approved for marketing June 8, 2026
- GenSci Pharmaceutical’s self-developed Class 1 new drug has been approved for clinical trials for two indications, covering pediatrics and neurology. June 8, 2026
- Yunnan Baiyao Group’s Central Research Institute’s INB301 injection IND has received FDA clinical trial approval, achieving a milestone breakthrough with “dual applications in China and the United States” June 8, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



