Eli Lilly wins another victory! Regcizosumab submits its marketing application in China
March 5, 2026
Source: drugdu
 122
122
 On March 3, the official website of the Center for Drug Evaluation (CDE) of the National Medical Products Administration of China showed that Eli Lilly and Company's application for marketing authorization of its Class 3.1 new drug, Lerechizumab injection, has been accepted.
On March 3, the official website of the Center for Drug Evaluation (CDE) of the National Medical Products Administration of China showed that Eli Lilly and Company's application for marketing authorization of its Class 3.1 new drug, Lerechizumab injection, has been accepted.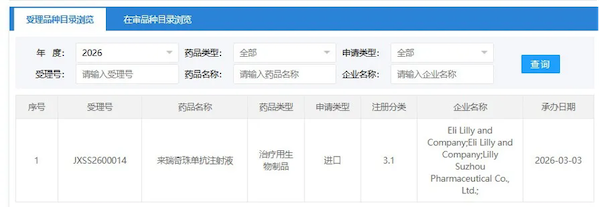 Lebrikizumab is an interleukin-13 (IL-13) inhibitor that selectively targets and neutralizes IL-13 with high binding affinity and a slow dissociation rate. The binding site of lebrikizumab to the IL-13 cytokine overlaps with the binding site of the IL-4Rα subunit in the IL-13Rα1/IL-4Rα heterodimer, thereby preventing the formation of this receptor complex and inhibiting IL-13 signaling. IL-13 is a major cytokine in atopic dermatitis, driving type 2 inflammation in the skin, leading to skin barrier dysfunction, itching, skin thickening, and infection.The development of lericizumab has been a long and winding road. Originally developed by Roche, the drug began its first clinical trial in 2008, but its initial indication of asthma did not go smoothly, resulting in a mixed record in two Phase III clinical trials in 2016. In August 2017, Roche licensed the drug to Dermira; in 2019, Dermira transferred its European options to Almirall; and in 2020, Eli Lilly acquired Dermira, gaining control of the product.In November 2023, leridgecizumab was first approved for marketing in Europe, followed by approval in the United States in September 2024, for the treatment of adolescents and adults aged 12 years and older with moderate to severe atopic dermatitis (AD). According to Eli Lilly's 2025 annual report, global sales of leridgecizumab were approximately $408 million. Eli Lilly holds exclusive rights to develop and commercialize leridgecizumab in the United States and all other regions of the world excluding Europe .The approval of lericizumab was primarily based on the results of the ADvocate 1, ADvocate 2 and ADhere studies, which primarily evaluated the clearance of skin symptoms in enrolled patients at 16 weeks.In the ADvocate 1 study, at week 16 of treatment, the proportions of subjects in the lerechizumab group and the placebo group who achieved an investigator global assessment score of 0 or 1 (IGA 0/1) with a decrease of at least 2 points from baseline were 43.1% and 12.7%, respectively (P<0.001), and the proportions of subjects who achieved an eczema area and severity index improvement of at least 75% (EASI-75) were 58.8% and 16.2%, respectively (P<0.001).In the ADvocate 2 study, at week 16 of treatment, the proportions of subjects achieving IGA 0/1 with a decrease of at least 2 points from baseline in the lerecizumab group and the placebo group were 33.2% and 10.8%, respectively (P<0.001), and the proportions of subjects achieving EASI-75 were 52.1% and 18.1%, respectively (P<0.001).Results showed that after 16 weeks of treatment, 38% of patients in the lerechizumab group achieved clear or near-clearance of skin symptoms (IGA 0 or 1), compared to 12% in the placebo group. In patients with clear or near-cleared symptoms, monthly dosing resulted in 77% maintaining remission for one year.In China, Eli Lilly registered a multicenter, randomized, double-blind, placebo-controlled phase III trial in 2024 to evaluate the efficacy and safety of lerechizumab in combination with or without topical corticosteroids in patients with moderate to severe atopic dermatitis. The primary endpoint was the proportion of patients achieving EASI-75 at week 16. Specific results have not yet been disclosed.Currently, only two IL-13 monoclonal antibodies have been approved for marketing globally: Tralokinumab (Leo Pharm) and Lelokinumab (Eli Lilly).
Lebrikizumab is an interleukin-13 (IL-13) inhibitor that selectively targets and neutralizes IL-13 with high binding affinity and a slow dissociation rate. The binding site of lebrikizumab to the IL-13 cytokine overlaps with the binding site of the IL-4Rα subunit in the IL-13Rα1/IL-4Rα heterodimer, thereby preventing the formation of this receptor complex and inhibiting IL-13 signaling. IL-13 is a major cytokine in atopic dermatitis, driving type 2 inflammation in the skin, leading to skin barrier dysfunction, itching, skin thickening, and infection.The development of lericizumab has been a long and winding road. Originally developed by Roche, the drug began its first clinical trial in 2008, but its initial indication of asthma did not go smoothly, resulting in a mixed record in two Phase III clinical trials in 2016. In August 2017, Roche licensed the drug to Dermira; in 2019, Dermira transferred its European options to Almirall; and in 2020, Eli Lilly acquired Dermira, gaining control of the product.In November 2023, leridgecizumab was first approved for marketing in Europe, followed by approval in the United States in September 2024, for the treatment of adolescents and adults aged 12 years and older with moderate to severe atopic dermatitis (AD). According to Eli Lilly's 2025 annual report, global sales of leridgecizumab were approximately $408 million. Eli Lilly holds exclusive rights to develop and commercialize leridgecizumab in the United States and all other regions of the world excluding Europe .The approval of lericizumab was primarily based on the results of the ADvocate 1, ADvocate 2 and ADhere studies, which primarily evaluated the clearance of skin symptoms in enrolled patients at 16 weeks.In the ADvocate 1 study, at week 16 of treatment, the proportions of subjects in the lerechizumab group and the placebo group who achieved an investigator global assessment score of 0 or 1 (IGA 0/1) with a decrease of at least 2 points from baseline were 43.1% and 12.7%, respectively (P<0.001), and the proportions of subjects who achieved an eczema area and severity index improvement of at least 75% (EASI-75) were 58.8% and 16.2%, respectively (P<0.001).In the ADvocate 2 study, at week 16 of treatment, the proportions of subjects achieving IGA 0/1 with a decrease of at least 2 points from baseline in the lerecizumab group and the placebo group were 33.2% and 10.8%, respectively (P<0.001), and the proportions of subjects achieving EASI-75 were 52.1% and 18.1%, respectively (P<0.001).Results showed that after 16 weeks of treatment, 38% of patients in the lerechizumab group achieved clear or near-clearance of skin symptoms (IGA 0 or 1), compared to 12% in the placebo group. In patients with clear or near-cleared symptoms, monthly dosing resulted in 77% maintaining remission for one year.In China, Eli Lilly registered a multicenter, randomized, double-blind, placebo-controlled phase III trial in 2024 to evaluate the efficacy and safety of lerechizumab in combination with or without topical corticosteroids in patients with moderate to severe atopic dermatitis. The primary endpoint was the proportion of patients achieving EASI-75 at week 16. Specific results have not yet been disclosed.Currently, only two IL-13 monoclonal antibodies have been approved for marketing globally: Tralokinumab (Leo Pharm) and Lelokinumab (Eli Lilly).
https://mp.weixin.qq.com/s/TMuqTf23nyCKb540QZpbXQ
Read more on
- A world first! Coagulation Factor X activator – Bomitai enzyme α for injection (Bojia Ning) – conditionally approved for marketing! June 10, 2026
- It’s here! Xanomeprazole Chloride Capsules (Kagero®) are now available, benefiting schizophrenia patients in China June 10, 2026
- World’s First! Tianguangshi’s High-Efficacy Drug Approved for Market Launch June 10, 2026
- Another powerful tool has been added to Qilu Pharmaceutical’s arsenal, with another blockbuster drug receiving market approval June 10, 2026
- Huadong Medicine’s application for a change of indication for its Yiyanshi® S-type forehead filler has been accepted, potentially marking another step forward in its high-end regenerative filler business. June 10, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



