IL-4R antibody submitted for market approval
February 27, 2026
Source: drugdu
 91
91
On February 25, the marketing application of Akeso Biopharma's IL-4R antibody mandocimab injection was accepted by the NMPA.
In August 2025, Akeso announced that its IL-4R antibody, mandocizumab, had achieved all efficacy endpoints in a Phase III clinical trial for the treatment of moderate to severe atopic dermatitis and planned to apply for market approval.
Based on inter-study comparisons (non-head-to-head), the efficacy data of mandocizumab is superior to that of the marketed product Dupixent, and it is being further expanded to adolescents.
Sanofi's IL-4R antibody, Dupixent, is the world's best-selling autoimmune drug, with sales of $18.5 billion in 2025.
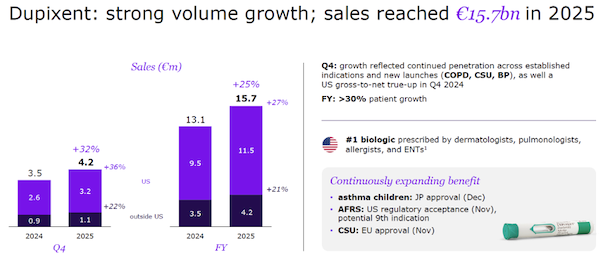 Image source: Medical Notes
Image source: Medical Notes
Conclusion
In the field of autoimmune diseases, Akeso Biopharma will focus on developing dual-target iterative products. The IL-4R/ST2 bispecific antibody AK139 is actively advancing its clinical development and has now reached the Phase II clinical trial stage.
https://news.yaozh.com/archive/47273.html
By editorRead more on
- The world’s first biased GLP-1RA officially enters commercialization. April 28, 2026
- Breaking News! BeiGene Introduces a PD-1/VEGF Triple Antibody April 28, 2026
- AstraZeneca’s dual immunotherapy has been approved for marketing in China for a new indication April 28, 2026
- A market worth hundreds of billions! Where is the next trump card in the cosmetic injection industry? April 28, 2026
- Thirteen Class 1 new drugs have been approved for clinical trials! Innovent, Hansoh, Gan & Lee, and Health-One, among others, have entered the fray April 28, 2026
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.



