-
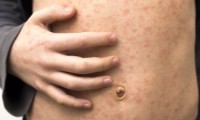
Rise in UK measles cases causing concern
- Source: drugdu
- 90
- May 8, 2023
-
New study reveals the best diets for a healthy heart
- Source: drugdu
- 85
- May 5, 2023
-
Mpox outbreak was wake-up call for smallpox preparation, vaccine maker Bavarian Nordic says
- Source: drugdu
- 62
- May 5, 2023
-
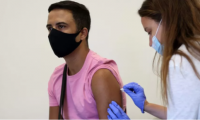
UK monkeypox vaccine rollout to wind down
- Source: drugdu
- 96
- April 28, 2023
-
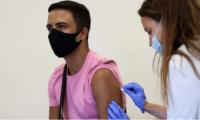
UK monkeypox vaccine rollout to wind down
- Source: drugdu
- 93
- March 30, 2023
-
US. Faces Severe Shortage of Some Drugs, Senate Report Says
- Source: drugdu
- 91
- March 23, 2023
-
Prioritize, Optimize And Modernize
- Source: https://invivo.pharmaintelligence.informa.com/IV124946/The-PostCOVID-Mantra-For-Medtechs-Prioritize-Optimize-And-Modernize
- 747
- April 6, 2022
-
FDA Approves First Treatment for COVID-19
- Source: drugdu
- 217
- October 26, 2020
your submission has already been received.
OK
Subscribe
Please enter a valid Email address!
Submit
The most relevant industry news & insight will be sent to you every two weeks.